Lecture 1
Units and significant figures
What will we learn in this course?
[Please read
the syllabus first.] In this course, we
will learn very deep principles of physics – Newton’s laws and conservation
principles. You will also learn how to
do lots of problems, but please try to appreciate the underlying big principles
beyond just the “how to” for each problem.
That way, you will get closer to the way physicists and scientists think
in general, and also you will score better grades.
Unit system
We will mostly
use the so-called “SI” unit system (le Système international
d'unités).
Its old name is the MKS unit system.
M is for meter (m), K for kilogram (kg), and S for second (s). These three units for length, mass, and time form
the sufficient basis of units for our course, and so they are called “base
units.” In general, you will need four
more base units (mole for amount, candela for luminosity, ampere for electric
current, and Kelvin for temperature), but we don’t need them in this course
(6A). The importance of base units is
that the unit of any arbitrary physical quantity can be expressed as a product
of powers of base units. For example, (as
we will learn soon) the SI unit of speed or velocity is m/s = m![]() , and the SI unit of energy is
, and the SI unit of energy is ![]() .
These units are examples of “derived units,” as opposed to base units. Some derived units have special names: Hz
(hertz) = 1/s, J (joules) =
.
These units are examples of “derived units,” as opposed to base units. Some derived units have special names: Hz
(hertz) = 1/s, J (joules) = ![]() .
.
Unit conversion
There
are quite a few unit systems in use, and thus unit conversions are
necessary. For example, here is a table
for converting some everyday units (or “English units”) such as pounds (lbs),
yard (yd), feet (ft), inches (in), and miles to and from appropriate SI units.
|
SI unit (w
prefix) |
E unit |
E unit, roughly |
Comments |
|
1 m |
3.28 ft, 1.09 yd,
39.4 in |
~ 3 ft, ~ 1 yd |
1 yd = 3 ft, 1 ft = 12 in |
|
2.54 cm |
1 in |
|
|
|
1 km |
0.621 miles |
~ 0.6 miles |
80 km |
|
1
m/s |
2.24
mph |
~
2 mph |
|
|
1 kg |
2.20 lbs |
~ 2 lbs |
|
|
1
rad |
57.3
o |
|
|
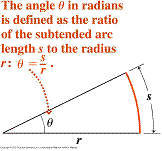 The last one deserves more
comments. The unit of angle in the SI
system is radian. The angle in radians
is defined as shown in this figure. So,
it follows that
The last one deserves more
comments. The unit of angle in the SI
system is radian. The angle in radians
is defined as shown in this figure. So,
it follows that ![]() radians = 360 degrees, since the circumference
of a circle is
radians = 360 degrees, since the circumference
of a circle is ![]() .
You will have to remember this if you
didn’t know it already.
.
You will have to remember this if you
didn’t know it already.
Also,
note that kg = 1000 g (=grams), cm = 0.01
m and km = 1000
m. Here, k (= kilo) and c (= centi) are
examples of unit prefixes (see
below).
You
should be able to convert units freely in this course. Unit conversion follows the general rules of
multiplication and division.
Example 1.
1 m = 3.28 ft. How many yards is
1 meter, given that 1 yd = 3 ft?
Answer: 1 yd = 3 ft means that 1
ft = ![]() yd. And
so,
yd. And
so, ![]()
Example 2.
Given that 1 km = 0.621 miles, how many mph (= miles per hour = miles/h)
is 1 m/s? Answer: Note that 1000 m = 1 km,
which means m = km/1000. Also note that 3600
s = 1 h, which means 1/s = 3600/h. Thus,
we get ![]() .
If you like, you can use clever
representations of 1, as follows. 1
km = 0.621 miles means 1000 m = 0.621 miles, which means 1 = 0.000621
miles/m. Also, 3600 s = 1 h means 3600
s/h = 1. Inserting these two clever
choices of 1, we get
.
If you like, you can use clever
representations of 1, as follows. 1
km = 0.621 miles means 1000 m = 0.621 miles, which means 1 = 0.000621
miles/m. Also, 3600 s = 1 h means 3600
s/h = 1. Inserting these two clever
choices of 1, we get ![]() .
Note that the symbol “m” appears twice, once in the numerator and once
in the denominator, canceling each other out.
The same goes for the symbol “s.”
Indeed, these canceling-outs were the purpose of the clever choices of
1. This way, we also get 1 m/s = 2.24
mph.
.
Note that the symbol “m” appears twice, once in the numerator and once
in the denominator, canceling each other out.
The same goes for the symbol “s.”
Indeed, these canceling-outs were the purpose of the clever choices of
1. This way, we also get 1 m/s = 2.24
mph.
Unit prefixes
We
already encountered kilo and centi. The
following table summarizes often used prefixes in this course (see textbook for
more). For example, 1 nm = ![]() m,
and 1 MHz =
m,
and 1 MHz = ![]() Hz
=
Hz
= ![]()
![]() .
.
|
Symbol |
G |
M |
k |
c |
m |
|
n |
|
Prefix |
giga |
mega |
kilo |
centi |
milli |
micro |
nano |
|
Power |
|
|
|
|
|
|
|
Note:
(1)
Case
matters! Often M (mega) and m (milli) seem
to be confused – they are different by 9 orders of magnitude!
(2)
Some
symbol is Greek! So, micro (![]() ) is distinct from milli
(m). Physicists seem to like Greek
symbols, and you will encounter a few of them in different contexts later in
this course. Among the “infamous” is
) is distinct from milli
(m). Physicists seem to like Greek
symbols, and you will encounter a few of them in different contexts later in
this course. Among the “infamous” is ![]() , which is omega. You will need to distinguish it from w.
, which is omega. You will need to distinguish it from w.
(3)
Presumably
you’ve heard about giga, mega, and kilo, in relation to computer memory. However, slight differences exist between
those computer science definitions and our physics definitions above.
Scientific notation
Any
non-zero real number can be written as
![]() where
where ![]() (“coefficient”)
is a real number satisfying
(“coefficient”)
is a real number satisfying ![]() and
and ![]() (“exponent”)
is an integer. We define this representation
as the “scientific notation.” [Strictly speaking, this is the “normalized” scientific notation,
where “normalized” means
(“exponent”)
is an integer. We define this representation
as the “scientific notation.” [Strictly speaking, this is the “normalized” scientific notation,
where “normalized” means ![]() . I.e., we take the convention
to always normalize for the scientific notation.] Often (e.g. in computer
codes)
. I.e., we take the convention
to always normalize for the scientific notation.] Often (e.g. in computer
codes) ![]() or
or ![]() is used to mean
is used to mean ![]() .
In this course, the notation
.
In this course, the notation ![]() may
sometimes be used to save space.
may
sometimes be used to save space.
Number of significant figures/digits
The
number of significant figures is defined as the number of digits used for the
coefficient ![]() in the scientific notation. So, significant digits are those digits that
serve the purpose other than
representing the order of magnitude (
in the scientific notation. So, significant digits are those digits that
serve the purpose other than
representing the order of magnitude (![]() ). Also, note that this definition leaves the #
of sig-figs of zero (e.g., 0.000) un-defined.
One should determine how to represent zero, by investigating the implied
error (see below).
). Also, note that this definition leaves the #
of sig-figs of zero (e.g., 0.000) un-defined.
One should determine how to represent zero, by investigating the implied
error (see below).
Example
3
![]() (# of sig-figs = 1),
(# of sig-figs = 1), ![]() (# of sig-figs = 4)
(# of sig-figs = 4)
![]() (# of sig-figs = 3) ,
(# of sig-figs = 3) , ![]() (# of sig-figs = 2)
(# of sig-figs = 2)
![]() (# of sig-figs = ambiguous; 1, 2 or 3?), -1001
= -1.001E3 (# of sig-figs = 4)
(# of sig-figs = ambiguous; 1, 2 or 3?), -1001
= -1.001E3 (# of sig-figs = 4)
![]() (# of
sig-figs = 1),
(# of
sig-figs = 1), ![]() (# of sig-figs
= 2),
(# of sig-figs
= 2), ![]() (# of
sig-figs = 3)
(# of
sig-figs = 3)
Note: Trailing zeros for integers (as in 100) are
always ambiguous, while trailing zeros appearing after the decimal point (as in
0.00100) are clearly significant. You
will see that many problems in the textbook do
use ambiguous expressions such as 100 kg.
By convention, we will take
this to mean three significant figures. In
general, most textbooks seem to automatically imply two or three significant
figures when numbers are written ambiguously.
Significant figures and errors
In
principle, all scientific numbers should be reported with error estimates, e.g.
the Bohr radius (roughly the radius of a hydrogen atom) is ![]() m. If all numbers are expressed in this way,
there is no need to consider “significant figures” at all. In other words, using the notion of
“significant figures” is a poor, but convenient, substitute for explicitly
specifying errors. The rough implied
error is determined by the rounding rule.
For instance, when the surface gravity is written as
m. If all numbers are expressed in this way,
there is no need to consider “significant figures” at all. In other words, using the notion of
“significant figures” is a poor, but convenient, substitute for explicitly
specifying errors. The rough implied
error is determined by the rounding rule.
For instance, when the surface gravity is written as ![]() , it implies that the error is
, it implies that the error is ![]() , where ~ means “roughly on the order of.”
, where ~ means “roughly on the order of.”
Rules of thumb for calculations involving significant figures
These
rough rules below are based on the principle that “the least accurate number
determines the overall accuracy.” [The virtue of these rules is that they are easy to
apply. However, if one were to use
explicit errors for all numbers, the error propagation theory must be used instead,
leading to a greater complexity. Thus, in essence, significant figures provide a quick and dirty
way to deal with errors and their propagation, but you would need to use
well-quantified error estimates whenever possible for your professional projects.]
A.
Adding (or subtracting) two
numbers. (i) Express the two numbers in the scientific
notation. (ii) If the two exponents differ, then
re-express the number with the smaller exponent by using the larger exponent,
which we will call ![]() .
In this conversion, the coefficient of the re-expressed number will be
made smaller than 1 in magnitude, and thus the re-expressed number will no
longer be in the scientific notation (i.e. not normalized any more), per our
definition above. Anyhow, at this point,
we have the two numbers expressed as
.
In this conversion, the coefficient of the re-expressed number will be
made smaller than 1 in magnitude, and thus the re-expressed number will no
longer be in the scientific notation (i.e. not normalized any more), per our
definition above. Anyhow, at this point,
we have the two numbers expressed as ![]() and
and ![]() . (iii) Now, we can add (or subtract) the two
coefficients,
. (iii) Now, we can add (or subtract) the two
coefficients, ![]() and
and ![]() , and let us call the result
, and let us call the result ![]() . Suppose
that
. Suppose
that ![]() has
has
![]() digits after the decimal point, and
digits after the decimal point, and ![]() has
has
![]() digits after the decimal point. Then,
you should round
digits after the decimal point. Then,
you should round ![]() so that its number of digits after the decimal
point is the smaller of
so that its number of digits after the decimal
point is the smaller of ![]() and
and ![]() . (iv)
Lastly, convert, if necessary, the result to the scientific notation.
. (iv)
Lastly, convert, if necessary, the result to the scientific notation.
B.
Multiplying (or dividing) two numbers.
If the two numbers have the # of sig-figs, ![]() and
and ![]() , then the result should be rounded so
that its # of sig-figs is the smaller of
, then the result should be rounded so
that its # of sig-figs is the smaller of ![]() and
and ![]() .
.
C.
Complicated
functions such as square root, sin, cos, … – the # of sig-figs remains
un-changed.
D.
For
multi-step calculations, keep at least one more sig-figs than that of the final
answer, in intermediate steps. This is to
prevent the accumulation of rounding errors.
[In reality, crunch out
numbers with your calculator with high machine precision, and keep the correct
number of sig-figs only at the end.]
Unfortunately,
the rule A is quite wordy and may seem complicated. This rule should not be forced into memory but
should be understood in terms of the underlying principle that “the least
accurate number determines the overall accuracy.” Examples should help.
Example 4
(1)
1.3
+ 0.001 = 1.3E0 + 1.0E-3 [step (i)] =
1.3E0 + 0.001E0 [step
(ii); now ready to add coefficients; 0.001E0 is not normalized] =
1.3E0 [step (iii); 1.3001 rounded to 1.3 since the initial error of the first term dominantly
determines the error of the outcome]
This example is so simple
that 1.3 + 0.001 = 1.3 would have been clear enough. Let us spell out the meaning of this result. 1.3 + 0.001 = (1.3 ![]() ) + (0.001
) + (0.001 ![]() ).
The 2nd term is clearly insignificant compared to the error
of the first term
).
The 2nd term is clearly insignificant compared to the error
of the first term ![]() , and so can be safely ignored.
, and so can be safely ignored.
(2)
3.70E2
+ 52 =
3.70E2 + 5.2E1 [step (i)] =
3.70E2 + 0.52E2 [step
(ii), now ready to add coefficients; 0.52E2 is not normalized] =
(3.70 + 0.52)E2 =
4.22E2 [3.70
and 0.52 have the same digits after the decimal point, and thus the same implied
errors, ![]() .
Steps (iii,iv) are unnecessary.]
.
Steps (iii,iv) are unnecessary.]
(3)
3.7E6
+ 5.2E5 [step
(i)] =
3.7E6 + 0.52E6 [step (ii)]
=
(3.7 + 0.52)E6 =
4.2E6 [step (iii); 4.22 rounded to 4.2 because of
3.7, whose error dominantly determines the error of the outcome]
Let us spell out the
last step. The sum 3.7 + 0.52 = (3.7
![]() ) + (0.52
) + (0.52 ![]() ) = (4.2
) = (4.2 ![]()
![]() ) + (0.02
) + (0.02 ![]() ).
As in (1), the 2nd term can be ignored to a good
approximation. Thus, “the least accurate number
determines the overall accuracy.”
).
As in (1), the 2nd term can be ignored to a good
approximation. Thus, “the least accurate number
determines the overall accuracy.”
(4)
3.789E6
– 5.1E3 [step (i)] =
3.789E6 – 0.0051E6 [step
(ii)] =
3.784E6 [step
(iii); 3.7839 rounded to 3.784 because of 3.789, whose error dominates in
determining the error of the outcome]
(5)
1.349E-2
– 1.2E-2 [step
(i)] =
0.1E-2 [step (iii); 0.149 rounded to 0.1 because of 1.2] =
1E-3 [step (iv); scientific notation]
(6)
3.0E8
![]() 2.16E-10 = (3.0
2.16E-10 = (3.0 ![]() 2.16) E-2 =
2.16) E-2 =
6.5E-2 [6.48
rounded to 6.5 (i.e. two sig-figs) because of 3.0]
(7)
2.16E8
/ 3E-10 = (2.16/3) E18 =
0.7E18 [0.72
rouded to 0.7 (i.e. one sig-fig) because of 3] =
7E17 [scientific
notation]
(8)
![]() [Keep the same # of sig-figs; rule C]
[Keep the same # of sig-figs; rule C]
(9)
![]() [Expect two sig-figs in the final result;
rules B,C] =
[Expect two sig-figs in the final result;
rules B,C] =
![]() [Keep at least one more sig-fig than
necessary in mid-step; rule D] =
[Keep at least one more sig-fig than
necessary in mid-step; rule D] =
1.9E2 [1.85 =
![]() ,
rounded to two sig-figs]
,
rounded to two sig-figs]
(10)
How
should you write 1.92 – 1.9? 0.02, 0.0
or 0?